臨床検査および血糖自己測定(SMBG)において、D-グルコースの測定に利用されます。
| 由来 | recombinant A. sojae |
|---|---|
| 系統名 | D-Glucose : acceptor 1-oxidoreductase |
| EC 番号 | 1.1.5.9 |
| 反応式 | D-Glucose + acceptor →→→ D-Glucono-1,5-lactone + reduced acceptor |

臨床検査および血糖自己測定(SMBG)において、D-グルコースの測定に利用されます。
| 由来 | recombinant A. sojae |
|---|---|
| 系統名 | D-Glucose : acceptor 1-oxidoreductase |
| EC 番号 | 1.1.5.9 |
| 反応式 | D-Glucose + acceptor →→→ D-Glucono-1,5-lactone + reduced acceptor |
| Appearance | yellow lyophilizate | |
|---|---|---|
| Activity | ≧ 700 U/mg lyophilizate | |
| Contaminants | NAD glucose dehydrogenase < 1.0×10-2 U/U% | |
| Hexokinase < 1.0×10-2 U/U% | ||
| α-glucosidase < 1.0×10-2 U/U% | ||
| β-glucosidase < 1.0×10-2 U/U% | ||
| Filterability | ≧ 90% | |
| Storage condition | below -20℃ |
| Molecular weight | ca. 85 kDa (SDS-PAGE) |
|---|---|
| Structure | monomer, one mole of FAD per mole of enzyme glycoprotein |
| Michaelis constant | 2.2×10-2 M (D-glucose) |
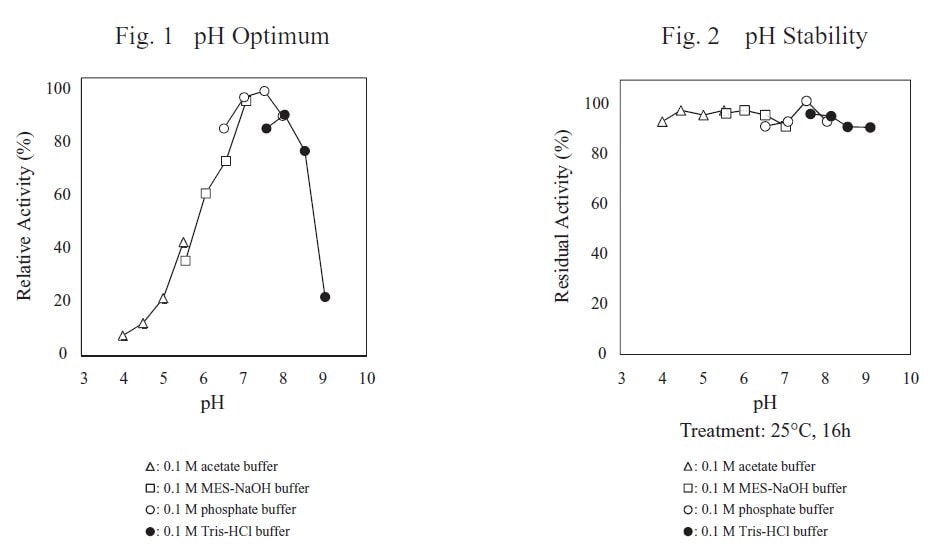
| pH Optimum | 7.0–7.5 |
| pH Stability | 4.5–8.0 |
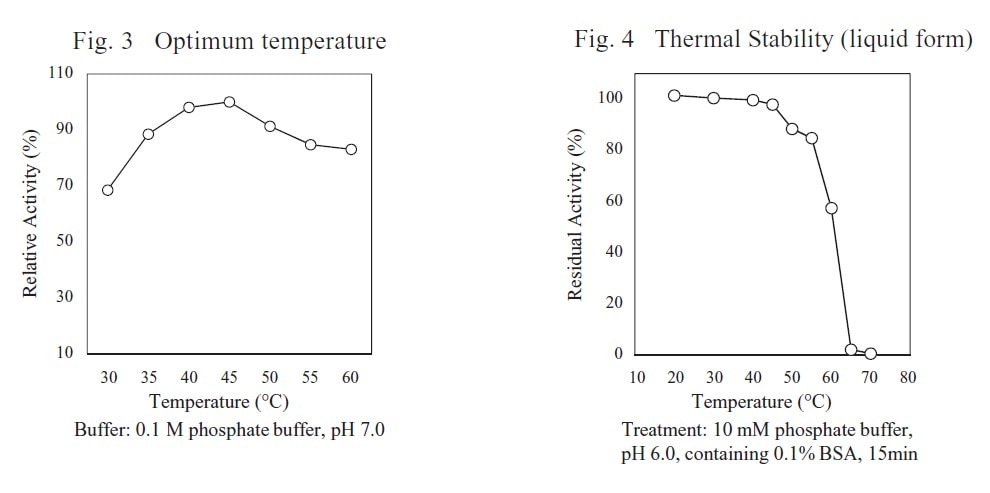
| Optimum temperature | 40–50℃ |
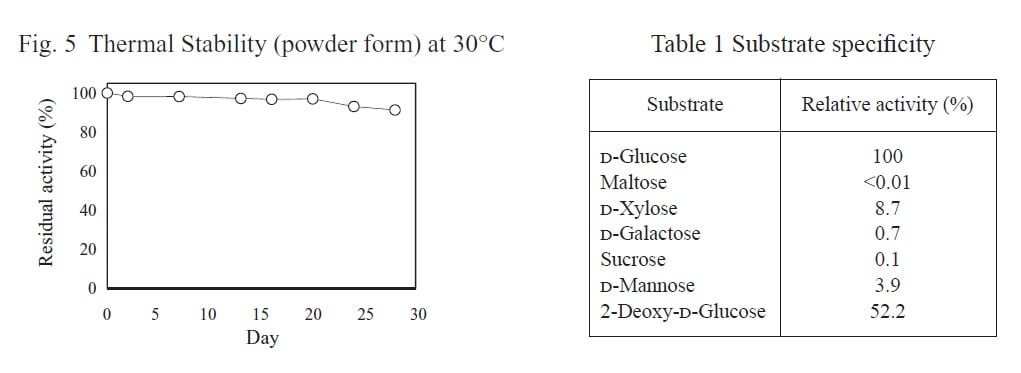
| Thermal stability | below 45℃ |
| Specificity | D-glucose (100), maltose (≺0.01), |
| D-xylose (8.7), D-galactose (0.7) , | |
|
sucrose (0.1) ,D-mannnose (3.9), 2-deoxy-d-glucose (52.2) |
The enzyme is useful for the determination of d-glucose in clinical analysis and self-monitoring blood glucose meters.

The disappearance of the blue color of DCIP by the reduction is measured spectrophotometrically at 600 nm.
One unit (U) causes the reduction of one micromole of DCIP per minute under the conditions described below.
Sample: dissolve the lyophilized enzyme to final concentration about 0.4 μg/mL with enzyme dilution buffer (ReagentE) immediately before measurement.
| 600 μL | D-Glucose solution | (Reagent A) |
|---|---|---|
| 2050 μL | Potassium phosphate buffer pH 7.0 | (Reagent B) |
| 150 μL | DCIP solution | (Reagent C) |
Activity can be calculated by using the following formula:
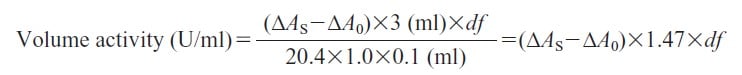
20.4 : Millimolar extinction coefficient of DCIP under the assay condition (cm2/μmol)
1.0 : Light pass length (cm)
df : Dilution factor
Hatada M, Loew N, Inose-Takahashi Y, Okuda-Shimazaki J, Tsugawa W, Mulchandani A, Sode K (2018)
Development of a glucose sensor employing quick and easy modification method with mediator for altering electron acceptor preference.
Bioelectrochemistry, 121, 185-190
Okuda-Shimazaki, J, Yoshida H, Sode K (2020)
FAD dependent glucose dehydrogenases – Discovery and engineering of representative glucose sensing enzymes –
Bioelectrochemistry, 132, 107414